
We’ll use a Bohr diagram to visually represent where the electrons are around the nucleus of the.  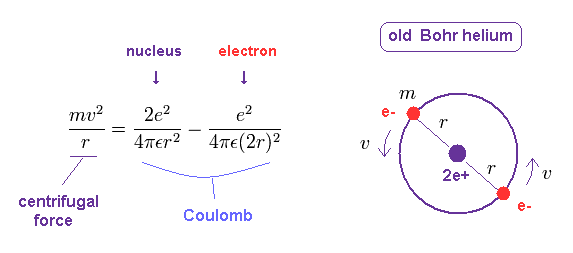
The orbit with n = 1 is the lowest lying and most tightly bound. In this video we'll look at the atomic structure and Bohr model for the Helium atom (He). In his final years, he devoted himself to the peaceful application of atomic physics and to resolving political problems arising from the development of atomic weapons.Īs n decreases, the energy holding the electron and the nucleus together becomes increasingly negative, the radius of the orbit shrinks and more energy is needed to ionize the atom. Proceeding from the double-cone model of Helium, based on Bohrs theorem and recently published in 13, a spherical modification could be made by introducing a second electron rotation which. (b) The energy of the orbit becomes increasingly less negative with increasing n.ĭuring the Nazi occupation of Denmark in World War II, Bohr escaped to the United States, where he became associated with the Atomic Energy Project. Therefore, it will not be valid for a singly ionised neon atom as it has. Thus the model simply throws out classical electromagnetic theory. Singly ionised neon atom (Ne+) Bohrs model is valid only for the single electronic systems. When an electron is in an 'allowed' orbit it does not radiate. It is the first atomic model based mainly on quantum mechanics. The model asserts that: The planetary model is correct. The Bohr model is a cake or planetary model of the atom, with electrons in shells. It comes to rest in the \(n = 6\) orbit, so \(n_2 = 6\).\): The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. The hydrogen atom model of Niels Bohr, published in 1913 1, and based on Max Planck’s quantum theory of light, published in 1900 2 and supplemented by Albert Einstein’s photoelectric. Then in 1913 Bohr, by accident, stumbled across Balmers numerology for the hydrogen spectrum, and in a flash came up with a workable model of the atom. In this case, the electron starts out with \(n = 4\), so \(n_1 = 4\). Bohr’s model of the hydrogen atom, proposed by Niels Bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. These last two equations can be used to calculate the radii of the allowed (quantized) electron orbits in any hydrogen-like atom. What is the energy (in joules) and the wavelength (in meters) of the line in the spectrum of hydrogen that represents the movement of an electron from Bohr orbit with n = 4 to the orbit with n = 6? In what part of the electromagnetic spectrum do we find this radiation? It is left for this chapter’s Problems and Exercises to show that the Bohr radius is. For example, an electrically neutral helium atom has an atomic number Z 2. Niels Bohrs model of the atom provided a wonderfully accurate explanation of the spectrum of hydrogen, but when it was applied to the spectrum of helium it failed.Werner Heisenberg developed a modification of Bohrs analysis but it involved half-integral values for the quantum numbers. 
The ratio of radii of first orbit of hydrogen atom and the second orbit of singly ionised helium atom will be. The energy of H atom in nth orbit is En, then energy in nth. Hydrogen-like ions are atoms of elements with an atomic number Z larger than one ( Z 1 for hydrogen) but with all electrons removed except one. The radius of first Bohr orbit in hydrogen atom is r0 then the radius of first orbit in helium atom will be. \): Calculating Electron Transitions in a One–electron System Bohr’s model of the hydrogen atom also correctly predicts the spectra of some hydrogen-like ions.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
